Save 10% off on your first order with coupon code: FIRST
Free with your purchase:

Purchase
Guarantee

Lowest Price
Guarantee

Identity Theft
Guarantee
Satisfaction Guarantee
A 30 day guarantee of up to a $1,000 of the purchase price, that you will receive the products/services you ordered, in accordance with the terms of sale.
Price Protection Guarantee
If the store's published price drops within 30 days of the purchase, you can make a claim for up to $100 of the difference.
ID Recovery Assistance
Provides help in the event of identity theft for up to 30 days after a sale, to help allay concerns about the security of your personal information. Up to total of $10,000 worth of specialist services.
Product Information
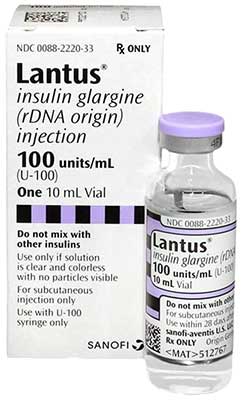
What is Lantus vial?
It is a human insulin analog invented to have low solubility at neutral pH. Production of Insulin glargine is through recombinant DNA technology of Escherichia coli.
Each mL of Lantus vial contains 100 units/mL of insulin glargine with a clear, colo\rless, aqueous solution. Sanofi-Aventis is the manufacturer and distributor of this drug.
What is it used for?
Lantus is a long-acting insulin used to maintain blood sugar levels in patients suffering from type 1 and type 2 diabetes mellitus and prevent diabetes complications. The doctor can prescribe this medication in combination with short-acting insulin.
How does it work?
Insulin glargine regulates glucose metabolism by inhibiting glucose production in the liver and stimulating peripheral glucose uptake in the muscle or fat. Lantus also enhances amino acid synthesis but inhibits lipolysis in adipocytes and proteolysis.
How to take it?
Only use this medication if it appears a clear, colorless, particle-free, aqueous solution. Keep the Lantus vial at room temperature for at least 1-2 hours before using it as it will be painful to inject it right away. Always use a new and sterile needle to avoid contamination. Wipe the top of the vial with alcohol wipes before injecting equal air to the insulin dose. Draw the dose by turning the vial and syringe upside down while holding it in one hand and pulling the plunger to withdraw the dose with another hand. Before removing the needle from the vial, check the inside of the syringe for air bubbles. Tap the syringe until the bubbles float on top and eject the air. Lantus can is administered via subcutaneous injection in the abdomen, deltoid, or thighs. Insulin may not cause an effect injected in a lumpy area. Always change the site of administration to lessen the risk of cutaneous amyloidosis and lipodystrophy. It is advised to take this medication at the same time once daily.
Dosage
Lantus is a prescription drug that must be ordered by a doctor for an individual. The factors to consider before determining the proper dose and timing are the lifestyle and blood glucose levels of the patient.
Side Effects
Dizziness, blurry vision, palpitations, headache, tremors, sweating, and slurred speech are the common side effects of Hypoglycemia in patients taking Lantus. Hyperglycemia can also be experienced due to insufficient insulin in the body to break down sugars. It may escalate to diabetic ketoacidosis or hyperosmolar coma.
Warnings, Precautions, and Contraindications
Propose glucose monitoring in patients taking Lantus as it may result in severe hypoglycemia. This drug is not intended for diluting with other insulin and administering it intravenously. Dose adjustments will be made for patients suffering from Liver and Kidney failure. Angiotensin Converting Enzyme Inhibitors (ACEIs), fibrates, Monoamine Oxidase Inhibitors (MAOIs), salicylates, sulphonamide antibiotics, fluoxetine, and disopyramide may enhance the lowering effect of blood sugar while corticosteroids, diuretics, glucagon, isoniazid, progesterone, thyroid hormones, danazol, epinephrine, clozapine, and salbutamol may reduce the lowering effect of blood sugar. Insulin glargine is contraindicated in patients with hypersensitivity to the excipients of Lantus.
Adverse Effect
Report it to the pharmacist or physician right away if the patient experience severe hypoglycemia, temporary visual impairment, Sodium retention, edema, Immune system disorders, taste disorders, and muscular pain.
Storage
An unopened Lantus vial is stored in a refrigerator (2-8°C) until expiration date. In-use Lantus vial must be stored in a room temperature not exceeding 30°C for a maximum of 28 days. Keep this medicine out of reach of children and pets. Do not freeze and expose to high temperatures.
Disposal
Do not discard Lantus in wastewater or household waste. Discard the used needles in a needle-resistant container labelled as “sharps”.