Save 10% off on your first order with coupon code: FIRST
Free with your purchase:

Purchase
Guarantee

Lowest Price
Guarantee

Identity Theft
Guarantee
Satisfaction Guarantee
A 30 day guarantee of up to a $1,000 of the purchase price, that you will receive the products/services you ordered, in accordance with the terms of sale.
Price Protection Guarantee
If the store's published price drops within 30 days of the purchase, you can make a claim for up to $100 of the difference.
ID Recovery Assistance
Provides help in the event of identity theft for up to 30 days after a sale, to help allay concerns about the security of your personal information. Up to total of $10,000 worth of specialist services.
Product Information
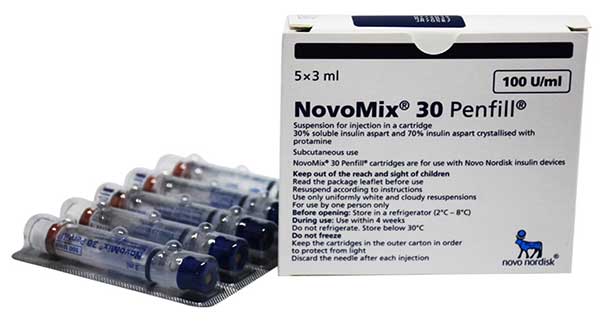
What is NovoMix 30 Penfill?
Is a biphasic suspension made from 30% soluble insulin aspart and 70% protamine-crystallized insulin aspart. Insulin aspart is produced in Saccharomyces cerevisiae via recombinant DNA technology.
A cartridge of NovoMix 30 PenFill contains 300 units/3mL. This drug is also available in another dosage form such as NovoMix 30 FlexPen.
What is it used for?
It lowers blood sugar levels after injection in people with Type 1 and Type 2 Diabetes Mellitus. NovoMix can be used alone or with other antidiabetic medicines.
How does it work?
It is a biphasic suspension that contains soluble insulin aspart that acts as a rapid-acting insulin analog and insulin aspart protamine that acts as an intermediate-acting insulin analog. This medication facilitates the uptake of glucose following the binding of insulin receptors on muscle and fat cells. It also inhibits glucose production in the liver.
NovoMix has a faster onset of action than regular human insulin. It starts to take effect within 10 to 20 minutes after administration. Maximum effect will occur between 1-4 hours and may last up to 24 hours.
How to take it?
NovoMix cartridge must be inserted into a Novo Nordisk Insulin delivery system. Take this medication via subcutaneous injection 10 minutes before meals. Make sure that the cartridge reaches room temperature before using it as it is easier to resuspend. Roll NovoMix cartridge 10 times horizontally between the palms then slowly move it up and down until it appears uniformly white and cloudy. The patient may administer this medication subcutaneously in the abdomen, thighs, upper arms, and buttocks. Rotate the injection site regularly to avoid the risk of developing lipodystrophy.
Dosage
The doctor will determine individual dosing for patients. Dose adjustment must be done to achieve optimal glycemic control.
Side Effects
Some signs and symptoms of Hypoglycemia are anxiety, headache, sweating, dizziness, confusion, mood change, and irritability are seen in patients taking NovoMix. Other common side effects are low potassium in the blood, itching, injection site reactions, peripheral edema, swelling in both hands and feet, and weight gain.
Warnings, Precautions, and Contraindications
NovoMix taken with ACE inhibitors, Fluoxetine, Fibrates, Sulfonamide antibiotics, Angiotensin II blockers, MAOIs, Salicylates, and other Antidiabetic agents may result in the risk of hypoglycemia. Glucose lowering effect of NovoMix may be lessened when co-administered with Corticosteroids, Progesterones, Diuretics, Atypical antipsychotics, Thyroid hormones, Estrogens, Glucagon, and Sympathomimetic agents. Dose adjustment must be done to closely monitor blood glucose levels in patients. Sharing NovoMix devices may pose a risk for the transmission of blood-borne pathogens. Do not drive or operate heavy machinery while on this medication.
Adverse Effect
Hypoglycemia is the most common adverse effect in patients taking NovoMix. Severe low blood sugar may result in temporary or permanent brain impairment, convulsions, unconsciousness, and death. Glucose monitoring is advised for all patients taking Insulin Aspart. During animal trials, there is no direct adverse effect on the fertility of male and female rats.
Storage
Novomix cartridges can be stored in the refrigerator while in-use cartridges must only be kept at room temperature for up to 4 weeks after first use.
Disposal
Put all used needles and cartridges in an FDA-cleared sharps disposal or a puncture-resistant container with a lid. Do not throw away needles and cartridges in the household trash. When the sharps disposal is full, follow the community guidelines in your area on how to discard sharps disposal containers.